Two main types of industrial radiation are used in industrial radiographic testing: X-rays and Gamma rays (γ). X-rays and gamma rays differ from other types of electromagnetic radiation only in their relatively short wavelengths that give these radiations the capability to penetrate material and expose internal defects.
As X-rays and gamma rays are physically similar, they differ only in how they are generated.
Principle of X-Rays
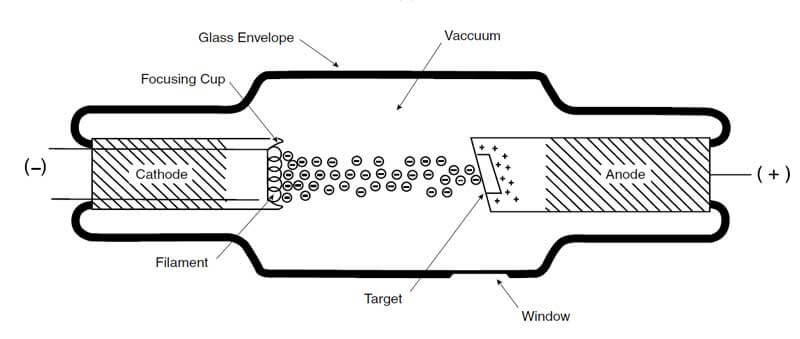
The X-ray tube consists of a glass envelope; the glass envelope, which is evacuated to an ultra-high vacuum, contains the anode (positive electrode) and the cathode (negative electrode).
Under the electrical tension effect (high voltage direct current) between the anode and the cathode, the cathode filament generates electrons, which get attracted to the anode and accelerate their speed. The voltage applied between the anode, and the cathode is called the X-ray tube voltage. The stream of electrons is concentrated into a beam by the focusing cup, and the surface area of the anode struck by electrons is called the target.
The accelerated electrons collide with a target on the anode. Their rapid motion is stopped, and a part of the electron’s energy converts to X-radiation. The X-radiation is known as X-rays, and the vacuum tube that utilizes this phenomenon is called an X-ray tube.
The target in the anode in most X-ray tubes is typically made of tungsten. The high atomic number of tungsten makes it an ideal material to interact with high-speed electrons. Besides, tungsten has a high melting point, making the target reliable to use under the high temperatures resulting from electron impingement. In fact, most of the kinetic energy from the electrons is converted into heat; therefore, the anode must have the capability to dissipate the heat. Typically, 97–99% of the electrons’ kinetic energy converts to heat, and approximately 1–3% converts to x-radiations.
The target size in the anode is an important parameter to consider; the target size directly affects the image sharpness of the object being radiographed and thus affects the definition of the final image. In general, the produced radiographic image is sharper and has a higher definition when the target size is small.
The total number of the generated electrons from the cathode can be adjusted by changing the X-ray tube’s filament voltage. Thus, it changes the dose of X-rays generated off the target of the anode. The change in X-ray tube voltage changes the speed at which electrons strike the target and, therefore, control the generated X-rays’ nature. The X-rays with relatively short wavelengths are called hard X-rays, while the X-rays that have relatively long wavelengths are called soft X-rays.
The radiation energy is expressed in voltage or kilo-voltage; however, the common expression for X-ray energy is kilo-electron volts (keV) or, as generally known, kilovolt (kV), which is equal to one thousand volts. Most industrial radiographic techniques use an energy range from about 100 kV to 400 kV. The intensity of the radiation is expressed in milliamperes (mA).
The radiation’s energy is known as the quality of the radiation, while the intensity of the radiation is known as the quantity of the radiation.
Principle of Gamma Rays
Gamma radiography is similar to X-radiography; the difference is how the radiation is generated. The significant advantage of gamma rays over X-rays is the compact size of gamma-ray sources, which allow gamma-ray shots to be performed in small and difficult to access places. The downside of using gamma rays is the longer exposure period than X-rays, and the gamma-ray source cannot be turned off like an X-ray.
The most common gamma-ray sources (radioactive isotopes) in industrial radiography applications are 192 and cobalt 60. Thulium 170, caesium 137, and radium 226 are also used in industrial applications but are limited.
Each radioactive isotope has a unique energy. Some of these radioactive isotopes give off a number of unique energy bundles. The radioactive isotope energy never changes; however, radioactive isotopes lose activity and decay with time. Decay means a reduction in activity or quantity, which varies depending on the kind of radioactive material. For instance, the intensity of the radiation from a cobalt 60 source decreases to half its original value in about five years; and that of an iridium 192 source in about 70 days. The amount of time it takes for a specific radioactive isotope to decay and reduce its activity to one half of its original is called the radioactive half-life.
The unit of radioactive isotope energy is electron volt (eV). A gamma-ray with an energy of 0.5 MeV (500 keV) is equivalent in wavelength and penetrating power to the most penetrating radiation emitted by an X-ray tube operating at 500 kV.
The amount of radiation given off by an isotope source known as the intensity is expressed in curies or becquerels. One curie is equal to 3.7 × 1010 disintegrations per second (decays per second), one becquerel is equal to one disintegration per second.
The degree of concentration of a radioactive source is known as the specific activity. Specific activity is expressed in terms of curies per gram. When comparing two gamma-ray sources of the same material and activity but different in the specific activity. The source with greater specific activity will be smaller in size. Therefore, the source of higher specific activity produces radiographic images with less geometrical unsharpness; besides, it will provide shorter source film distances and shorter exposure time.
Difference between X-Rays and Gamma Rays
As previously mentioned, X-rays and gamma rays are physically similar; however, they differ only in how they are generated and their material properties.
The main difference between the two radiations sources are as follow:
- X-rays require electric power to operate, while gamma rays require no electric power supply, making it easy to use in the field. However, an X-ray source from the safety perspective upon disconnecting the power source will be switched off and have no associated risk, while a gamma-ray source cannot be switched off.
- Gamma rays can be obtained in a range of source diameters; thus, they can be used with a very short source-to-film distance and a small diameter source.
- Gamma rays provide a wide variety of radiation hardnesses (energy level); however, the energy level (radiation hardness) and intensity cannot be adjusted. On the opposite side, adjusting the energy level as well intensity is possible with X-rays.
- Gamma rays have higher radiation hardness (more penetration power) than those of conventional X-ray equipment can produce.
- X-rays produce higher radiographic image contrast than gamma rays.
- Gamma rays have a limited service life due to source deterioration.
- Gamma-ray sources are compact, which allows the test to occur in small and difficult to access places.
Read: What Are The Certification Programs Of NDT Personnel?
References: